Enantioenriched dienols are not only widely present in natural products and pharmaceutical molecules as important bioactive structural motifs, but also serve as key building blocks in organic synthesis. Consequently, the development of general and efficient synthetic methods for chiral conjugated dienols has remained an important goal in synthetic chemistry. Traditionally, these compounds have mainly been prepared through the addition of preformed alkenylmetal reagents to aldehydes, but such approaches are often operationally cumbersome and suffer from poor atom economy. Although reductive coupling strategies can generate alkenylmetal species in situ from alkenyl halides or alkynes in the presence of reductants, they still face limitations such as unsatisfactory atom economy. By contrast, the direct use of simple 1,3-dienes as nucleophiles in additions to aldehydes would undoubtedly provide a more ideal strategy for the synthesis of chiral conjugated dienols. However, realizing such asymmetric direct addition reactions remains highly challenging in terms of the design and development of effective catalytic systems.
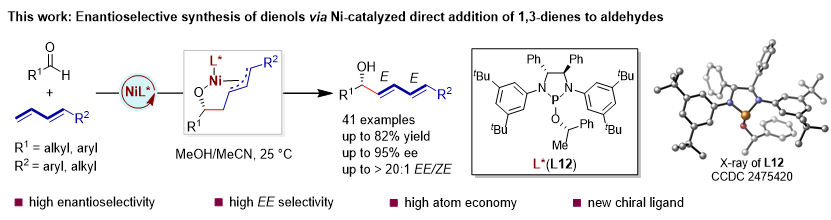
Figure 1. Nickel-catalyzed asymmetric direct addition of 1,3-dienes to aldehydes.
Recently, the Li-Jun Xiao group designed and synthesized a new class of diamidophosphite ligands based on chiral diamine scaffolds, and, for the first time, realized a nickel-catalyzed asymmetric direct addition of 1,3-dienes to aldehydes, thereby providing a new method for the efficient and atom-economical synthesis of chiral dienols (Figure 1). This method exhibits not only excellent regioselectivity and enantioselectivity, but also mild reaction conditions and broad functional-group compatibility. Notably, these newly developed chiral diamidophosphite ligands are readily accessible and structurally tunable, and thus are expected to find broad applications in a wider range of asymmetric catalytic transformations. Relevant achievements were published in Angew. Chem. Int. Ed., 2026, DOI: 10.1002/anie.2370635.