Research on pKa values constitutes an important area of benchmark data studies in chemistry. In gas-phase chemistry, under the ideal vacuum conditions free from medium interference, the pKa value corresponding to the deprotonation dissociation equilibrium constant of an X–H bond in an organic compound can be defined as the “absolute pKa”. However, gas-phase chemical studies currently suffer from many limitations and practical inconveniences, and the amount of available data on gas-phase absolute pKa values remains extremely limited. Moreover, since the vast majority of organic chemical reactions require the participation of solvents, it is necessary to establish a benchmark thermodynamic data system referenced to a specific solvent and, through experiment, to construct an “absolute pKa scale in solution.” This concept was introduced in 1975 by the renowned physical organic chemist and U.S. National Academy of Sciences member Professor Frederick G. Bordwell, who defined the experimentally determined pKa scale in DMSO, based on the self-deprotonation equilibrium constant of a specific anchor compound, as the “absolute acidity scale” in DMSO.[1] In 2012, the distinguished physical organic chemist and Chinese Academy of Sciences member Jinpei Cheng, in his well-known article on the standard pKa scale in ionic liquids, reiterated the definition of the “absolute pKa scale in solution”: “If the self-deprotonation equilibrium constant of a suitable indicator can be directly measured in solution without the need to add a base to establish equilibrium, then this pKa value can be defined as an absolute pKa value in solution, and can further serve as a reference point for constructing the absolute pKa scale in that solvent.”[2] Not long ago, with the support of Professor Jinpei Cheng, the research group of Tianfei Liu at Nankai University developed a brand-new anchor compound, 2-(4-trifluoromethanesulfonylphenyl)propanedinitrile. The group experimentally determined the self-deprotonation equilibrium constants of this anchor compound in six polar fluorinated aromatic solvents, including fluorobenzene, difluorobenzene, and trifluoromethylbenzene, and on this basis experimentally established the absolute pKa scale in solution for a series of C–H acidic compounds in these solvents.[3]
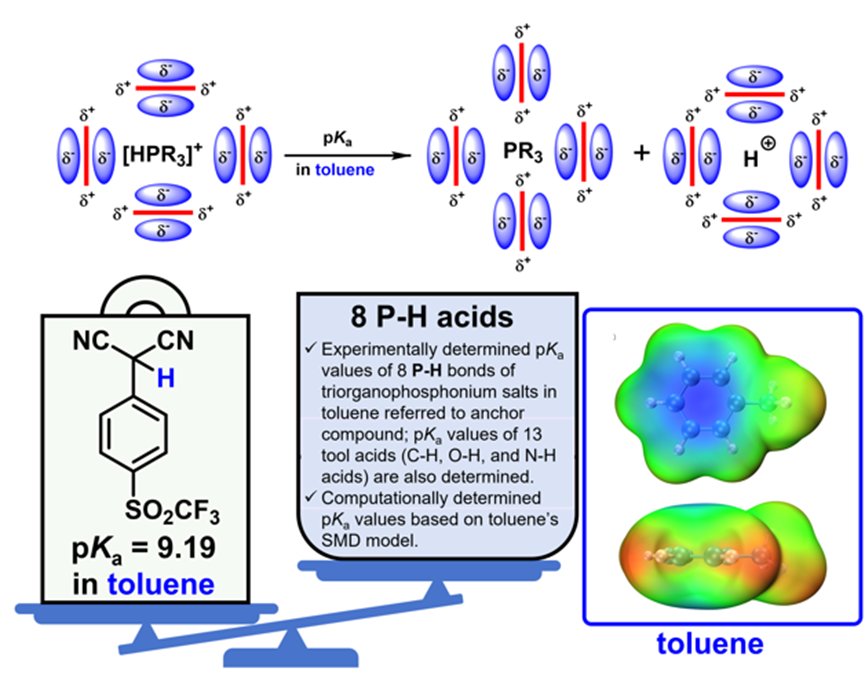
In solution, the acid–base strength of a solute is affected by the solvent’s own solvent basicity as well as various nonspecific interactions between the solvent and the solute. Notably, for toluene, a low-dielectric, aprotic solvent, its inherently weak solvent basicity makes it highly challenging to experimentally determine the self-dissociation constant of an anchor compound with clear physicochemical significance. As a result, the absolute pKa scale in solution for toluene, a solvent widely used in organic synthetic chemistry and organometallic chemistry, has long remained an unexplored frontier. For nearly the past three decades, synthetic chemists and organometallic chemists have had to rely on pKa values measured in other solvents to guide their studies in toluene solution systems. Recently, Yunfei Liang and other members of Tianfei Liu’s research group experimentally determined for the first time the self-dissociation constant in toluene of the anchor compound 2-(4-trifluoromethanesulfonylphenyl)propanedinitrile developed by the group. The absolute pKa value in solution of this compound in toluene was found to be 9.19.[4] Using this value as a benchmark, Tianfei Liu’s group successfully established, through experiment, the absolute pKa scale in solution in toluene for 13 reference compounds, including 10 C–H acids, as well as 8 organophosphine bis(trifluoromethanesulfonyl)imide salts, by means of UV–visible absorption spectroscopy and 31P{1H} NMR spectroscopy.[4]These experimentally determined pKa values show an excellent linear correlation with the DFT-calculated values based on the SMD solvation model for toluene, thereby validating the accuracy and reliability of the experimental data. This represents the first report of an absolute pKa scale in solution with clear physicochemical significance in toluene. The pKa values experimentally determined in this work for toluene can provide important guidance for organic synthesis, catalytic reactions, and organometallic reactions.
This work was carried out with the support of the State Key Laboratory of Elemento-Organic Chemistry at Nankai University, the Frontiers Science Center for New Organic Matter, and the Haihe Laboratory of Sustainable Chemical Transformations. Financial support for this work was provided by the National Key R&D Program of China, the National Natural Science Foundation of China, the Tianjin Special Fund for State Key Laboratories, and the Fundamental Research Funds for the Central Universities.
Reference:
[1] W. S. Matthews, J. E. Bares, J. E. Bartmess, F. Bordwell*, F. J. Cornforth, G. E. Drucker, Z. Margolin, R. J. McCallum, G. J. McCollum, N. R. Vanier, J. Am. Chem. Soc. 1975, 97, 7006−7014.
[2] H. Deng, X. Li, Y. Chu, J. He, J.-P. Cheng*, J. Org. Chem. 2012, 77, 7291−7298.
[3] X. Kong, Y. Liang, Z. Guo, T. Lin, S. Liu, Z. Liu, T. Liu*, J.-P. Cheng, ChemSusChem, 2025, 18, e202402041.
[4] Y. Liang, X. Kong, T. Lin, Z. Guo, T. Liu*, J. Org. Chem., 2026, 91, 3941–3949.