ER2 is an important transmembrane receptor tyrosine kinase and a key member of the epidermal growth factor receptor (EGFR) family. It is highly expressed in a variety of tumors, including breast cancer. Through homo- or heterodimerization involving its extracellular domain and transmembrane region, HER2 mediates aberrant activation of downstream signaling pathways, thereby promoting tumor cell proliferation and metastasis. In recent years, although techniques such as protein crystallography and cryo-electron microscopy (Cryo-EM) have revealed structural features of the HER2 extracellular domain (ECD), kinase domain, and antibody binding, there is still a lack of direct biochemical and structural evidence to clarify the conformational characteristics of the HER2 transmembrane region and whether antibody binding induces dynamic changes in this region.
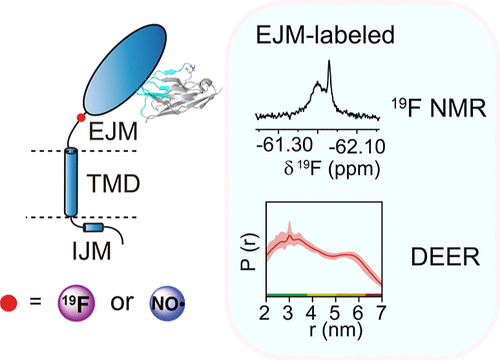

Recently, the research group of Chan Cao at Nankai University developed an efficient strategy for constructing phospholipid nanodiscs with site-specific intramembrane labeling, termed NMPCR, and successfully established a HER2 model system with site-specific labeling at different intramembrane positions in a near-native membrane environment. High-sensitivity 19F NMR chemical probes suitable for membrane protein studies were designed and compared. By combining protein site-specific labeling chemistry, 19F NMR, and PD-EPR, the dynamic features of the transmembrane region and juxtamembrane domain of the HER2 transmembrane receptor in a phospholipid bilayer were systematically characterized, and the effects of antibody binding on the dynamic behavior of key sites in the transmembrane region were identified. The study found that the extracellular domain of HER2 exerts a significant conformational constraint on the transmembrane-juxtamembrane region, and that antibody binding can trigger marked conformational changes in the juxtamembrane region, providing new structural biological evidence for understanding the mechanism of HER2 signal transduction. This work provides a new perspective for studying the dynamic mechanism of the HER2 transmembrane region and reveals, for the first time, the long-range regulatory effects of the HER2 extracellular domain and antibody binding on the conformation of the transmembrane region. These findings not only deepen the understanding of the molecular mechanism of HER2 signal transduction and provide important theoretical support for the development of HER2-targeted cancer therapies, but also offer technical support for studies on the dynamic properties of other similar receptors. Relevant achievements were published in J. Am. Chem. Soc., 2026. DOI: 10.1021/jacs.5c11966.