Cytochalasans are a large class of polyketide natural products featuring an isoindolinone structural unit. Because they can bind to intracellular actin and inhibit its polymerization, cytochalasans have long been widely used as small-molecule tools for studying the structure of the cytoskeleton and its dynamic regulation. Meanwhile, this family of natural products also exhibits diverse biological activities, including immunomodulatory effects, cytotoxicity, and nematicidal activity. In 2018, Professor Jikai Liu’s group isolated two new cytochalasan natural products—Curtachalasin A and Curtachalasin B—from an endophytic fungus parasitic on potato. Unlike the commonly encountered (5/6/11) tricyclic scaffold of traditional cytochalasans, these two molecules, for the first time, presented an unprecedented pyrrolidine/perhydroanthracene-type (5/6/6/6) tetracyclic framework. (Figure 1).
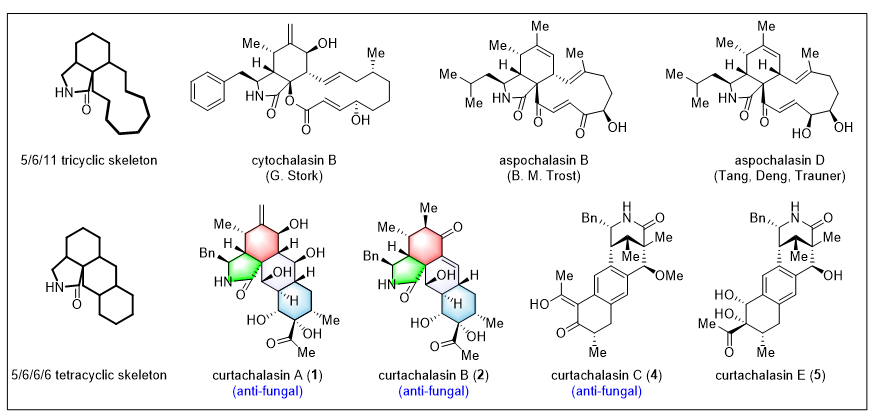
Figure 1. The structure of curtachalasin B
Recently, based on a bioinspired synthetic strategy, Professor Jun Deng’s group at Nankai University developed a modular and scalable approach, accomplishing the first total synthesis of Curtachalasin B and, in parallel, the synthesis of 14 related family members.The key innovations of this work lie in: (1) modular convergent synthesis combined with late-stage methyl addition, enabling efficient access to the key precursor zygosporin G; and (2) a semi-pinacol rearrangement together with a B(C6F5)3-promoted transannular cyclization/α-ketol rearrangement cascade, which biomimetically constructs the complex tetracyclic framework. This study not only provides a broadly applicable synthetic strategy for curtachalasin-type natural products, but also offers direct chemical evidence, at the molecular level, for their biosynthetic pathway. Relevant achievements were published in Angew. Chem. Int. Ed., 2026, DOI: 10.1002/anie.202524740.